(in Polish) Przedmioty minimum programowego dla studentów 3-go semestru (S1-CH) (course group defined by Faculty of Chemistry)
|
Key
If course is offered then a registration cart will be displayed.
 - you are not logged in
- you are not logged in 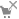 - currently you are not allowed to register
- currently you are not allowed to register  - you are allowed to register
- you are allowed to register  - you are allowed to unregister (or withdraw application)
- you are allowed to unregister (or withdraw application) 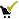 - you applied for registration (and you cannot widrdraw this application)
- you applied for registration (and you cannot widrdraw this application) 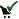 - you are registered (and you cannot unregister)
- you are registered (and you cannot unregister)
Use one of the "i" icons below for additional information.
2023Z - Winter semester 2023/24 2024Z - Winter semester 2024/25 (there could be semester, trimester or one-year classes) |
Actions | |||||||
|---|---|---|---|---|---|---|---|---|
| 2023Z | 2024Z | |||||||
| 1200-1CHANALC3 |
 
|
 
|
Classes
Winter semester 2023/24
Groups
Brief description
Calculations related to classic methods: gravimetric and volumetric – titrations based on the following reactions: acid-base, precipitation, reduction and oxidation, complex formation. |
|
||||
| 1200-1CHANALAW3 |
 
|
 
|
Classes
Winter semester 2023/24
Groups
Brief description
o acquaint students with fundamentals of classical quantitative analysis (titrations) |
|
||||
| 1200-1CHANALAL3 |
 
|
 
|
Classes
Winter semester 2023/24
Groups
Brief description
The aim of laboratory is to present classic methods of quantitative analysis applied in practice in many laboratories among other connected with environmental protection. Individually performed titrations based on: acid-base, precipitation, reduction and oxidation, complex formation reactions. The manual experience acquired is essential in laboratory work for every chemist. Laboratory is accurately connected with laboratory „ Introduction to Instrumental Analysis” performed during IV semester. |
|
||||
| 1200-1CHF1W3 |
 
|
 
|
Classes
Winter semester 2023/24
Groups
Brief description
The physical chemistry course is to lay foundations in thermodynamics, thermochemistry, chemical equilibrium, electrochemistry, chemical kinetics and to present how to use in practice the laws describing physicochemical phenomena. These fundamental laws are then used In other branches of chemistry: organic and analytical chemistry, technology and biochemistry. |
|
||||
| 1200-1CHF1C3 |
 
|
 
|
Classes
Winter semester 2023/24
Groups
Brief description
The student will get knowledge on how to solve mathematical problems within two (out of four) basic sub-fields of physical chemistry. Self-paced work on selected problems is necessary. |
|
||||
| 1200-1CHFIZ1AL3 |
 
|
 
|
Classes
Winter semester 2023/24
Groups
Brief description
Laboratory experiments are related to the essential sections of the contents of the physical chemistry I lecture. The students work in two person groups under supervision of a teaching assistant. 10 (level B) or 6 (level A) experiments are to be completed by every group. The experiments are divided into two thematic sections: I – thermodynamics and phase equilibria, II – termochemistry and properties of the matter. In every section the experiments are strictly related to the scientific topic of the section. Time of a single experiment – 5 hrs. |
|
||||
| 1200-1CHOR1A3 |
 
|
 
|
Classes
Winter semester 2023/24
Groups
Brief description
Lecture The properties, methods of preparation and reactivity (with particular emphasis on reaction mechanisms) of organic compounds with specific functional groups are thoroughly discussed. Seminar With the participation of the student, the properties of organic compounds, their stereochemistry, resonance structures and the reaction mechanisms are discussed. |
|
||||
| 1200-1FIZL3 |
 
|
 
|
Classes
Winter semester 2023/24
Groups
Brief description
The purpose ot the lab is the familiarisation with fundamental physical laws and phenonomena through conducting measuments, analysing the obtained data and preparing a report of the conducted experiment. |
|
||||
| 1200-1PCHTW3 |
 
|
 
|
Classes
Winter semester 2023/24
Groups
Brief description
Explanation of basic concepts used to a description of electronic structure of atoms and molecules by methods of quantum chemistry |
|
||||
| 1200-1PCHTL3 |
 
|
 
|
Classes
Winter semester 2023/24
Groups
Brief description
Using simple quantum chemical models to describe properties of molecules and chemical reactions: theory and computational practice. Modeling relations between microscopic structure and properties of matter. |
|
||||